 What Courses Can I Take to Satisfy Joshua’s Law?. Featuring a brushed gold-finish metal base and a glass shade, this table lamp … Add some cool, retro-style charm to your lighting with this 21" H Table Lamp from Joss & Main. By Abbey White Ap10:56am Drake Bell, former Nickelodeon star and musician, has been found. The former 'Drake & Josh' star had last been seen on Wednesday night in Florida. An example of an unreasonable answer would be if you got a negative number since negative pressures don’t make sense.Drake Bell Reported “Missing and Endangered” by Daytona …. 
Also, don’t forget the negative sign in front of the heat of vaporization! Keep an eye on your units!Īs we solve, the heat of vaporization and the R constant have different units. Now we plug in our known values and solve. Then, to make it easier we multiply both sides by -1 to get rid of the negative sign on P 2. Therefore, our strategy will be to rearrange and then plug into the equation to solve. Looking at our equations, all but one variable is known. We only have the temperature for the other point, 313 K (40 oC). One temperature and pressure point is 296 K (23 oC) and 120 mmHg. What is the vapor pressure at elevated temperature for the liquid?įirst, let’s layout the variables we know. The heat of vaporization of the liquid is 48.2 KJ/mol. You know at 23 oC it had a vapor pressure of 120 mmHg. Suppose you have a liquid that you heat to 40 oC. So, at the end, we need to convert units. Make sure to always check your units! For example, here R has units of joules but the question asked for units of kilojoules. 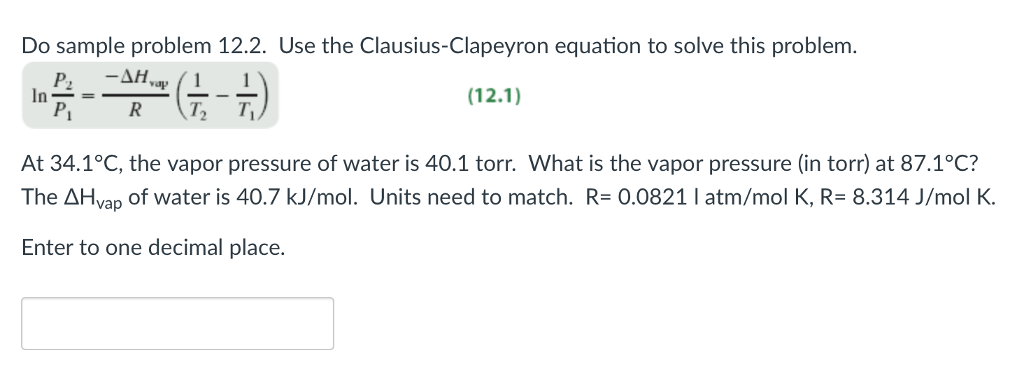
Plug these values into our equation above: We know P 2 because the boiling point will be at the standard pressure, which is 760 mmHg. Our second P 2 and T 2 are 760mmHg and 314 K (41 oC). 
The temperature needs to be in kelvin because our R constant has units of Kelvin. Our P 1 and T 1 will be 400 mmHg and 295 K (22 oC). We have the equation from above that we can easily plug our two known points into and solve for. We could solve the problem either graphically or algebraically. What is the heat of vaporization in KJ/mol? Given a liquid that has a normal boiling point of 41.0 oC and a P vap= 400 mmHg at 22 oC. Clausius Clapeyron Equation Example Problems Calculating Heat of Vaporization The equation can then be rearranged into the other forms to easily solve for whatever variable you need. The equation comes from the fact that at both temperatures, the constant C (from the very first equation) must be equal. ΔH vap is the enthalpy of vaporization for the substance. In these equations, P 1 and P 2 are the vapor pressures at T 1 and T 2. This is done by plugging in two known vapor pressures and temperatures to either of the equations below. The slope of the line will be the vapor pressure.Īnother technique is to determine algebraically what the heat of vaporization is. The vapor pressure and temperature can then be plotted. 
To determine the heat of vaporization, measure the vapor pressure at several different temperatures. The equation can be used to solve for the heat of vaporization or the vapor pressure at any temperature. Other names for the equation include the Clapeyron equation or the Clapeyron-Clausius equation. To make the graph linear, the natural log of the vapor pressure verse temperature is plotted. And C is a constant that is specific to the liquid being examined.īelow is a plot of the vapor pressure of water verse the temperature. Vapor pressure rises non-linearly as temperature rises since the y-term in this case is a natural log of the vapor pressure. The Clausius Clapeyron equation is shown below in a form similar to a linear equation (y=mx+b). The Clausius-Clapeyron equation relates the heat of vaporization and vapor pressure. At the end, there are several problems with step-by-step solutions. Core Concepts: In this tutorial, you will learn what the Clausius Clapeyron Equation is and how to apply it to problems. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
